Zonisamide audit report
Zonisamide is a new generation antiepileptic drug licensed in the UK for adjunctive treatment of refractory partial-onset seizures with or without secondary generalisation. This audit aims to evaluate the efficacy and tolerability of zonisamide experienced by patients in our service at Barts and The London NHS Trust. Data was gathered from nine patient files which demonstrated low efficacy and high treatment discontinuation rates compared with large studies though a similar side effect profile. The small size of the audit negates large conclusions; however, the distinct lack of efficacy of zonisamide in our patients should be considered in future management decisions.
- Barts and The London NHS Trust, Department of Neuroscience.
- Clinical Lead: Dr Cockerell, Clinical Director of Clinical Neurosciences and ENT Clinical Academic Unit at Barts and The London NHS Trust.
- Project Lead / Report written by: Sean Ferriter, 4th year medical student at Barts.
- Date: May 2010.
Background
Epilepsy is a chronic neurological disorder associated with significant impairment. The prevalence of active epilepsy is 5-10/1000, with approximately 50 million people affected worldwide. Even with our modern pharmacopeia, 30% of epileptics have poorly controlled seizures.5,20
Epilepsy can be classified as partial or generalised depending on the distribution of paroxysmal cerebral neuronal discharge. Partial epilepsy accounts for 60% of cases, and tends to be more difficult to treat than generalised. Attaining satisfactory control is paramount given the serious and pervasive sequelae of uncontrolled epilepsy: reduced quality of life, impaired cognition, increased mortality (including sudden unexpected death in epilepsy).2,18
Following diagnosis, ideal management takes the form of monotherapy. If this proves unsuccessful in seizure control an alternative monotherapy is attempted and administered by titrating up the new drug and tapering off the initial drug simultaneously. Refractory epilepsy requires polytherapy with adjunctive medication as the multiple mechanisms of action can contribute to an overall antiepileptic effect not possible from the action of a single drug. The abundance of new generation antiepileptic drugs (AEDs), together with the older AEDs provides a wide range of possible combinations.9,15
This audit was conducted to evaluate the place of zonisamide, a new AED, in the epilepsy service run at Barts and The London NHS Trust, given that practice guidelines for polytherapy with new generation AEDs remain empirical.12
Epilepsy is a chronic neurological disorder associated with significant impairment. The prevalence of active epilepsy is 5-10/1000, with approximately 50 million people affected worldwide. Even with our modern pharmacopeia, 30% of epileptics have poorly controlled seizures.5,20
Epilepsy can be classified as partial or generalised depending on the distribution of paroxysmal cerebral neuronal discharge. Partial epilepsy accounts for 60% of cases, and tends to be more difficult to treat than generalised. Attaining satisfactory control is paramount given the serious and pervasive sequelae of uncontrolled epilepsy: reduced quality of life, impaired cognition, increased mortality (including sudden unexpected death in epilepsy).2,18
Following diagnosis, ideal management takes the form of monotherapy. If this proves unsuccessful in seizure control an alternative monotherapy is attempted and administered by titrating up the new drug and tapering off the initial drug simultaneously. Refractory epilepsy requires polytherapy with adjunctive medication as the multiple mechanisms of action can contribute to an overall antiepileptic effect not possible from the action of a single drug. The abundance of new generation antiepileptic drugs (AEDs), together with the older AEDs provides a wide range of possible combinations.9,15
This audit was conducted to evaluate the place of zonisamide, a new AED, in the epilepsy service run at Barts and The London NHS Trust, given that practice guidelines for polytherapy with new generation AEDs remain empirical.12
Manufacture
Zonisamide, brand name Zonegran, is a new generation antiepileptic drug. It was discovered in 1972, subsequently launched in Japan in 1989, the USA in 2000, and within the EU in 2005. Originally launched by Dainippon Sumitomo Pharma, but taken over by Elan, and then by Eisai.16,19-21
Indications
The European Medicines Agency (EMEA) in Europe and the Food and Drug Administration (FDA) in the USA have approved the use of zonisamide as adjunctive therapy in adults with refractory partial-onset seizures with or without secondary generalisation.22
Japan has a much broader licence including most seizure types, use as adjunctive therapy or monotherapy, and use in adults and children.13
UK/USA off-label use in epilepsy is for a similar profile as the Japanese licence.22 Off-label and experimental uses of zonisamide include for Parkinson’s disease, resting tremor, essential tremor, obesity (eating disorders – particularly binge eating), migraine prophylaxis, and psychiatric disorders.22
Mechanism of Action
Zonisamide is a benzisoxazole-derived sulphonamide anticonvulsant structurally distinct from other AEDs and has multiple mechanisms of action contributing to its effectiveness across seizure types.10,21 Blockage of voltage-gated sodium channels is thought to account for its effect against tonic-clonic and partial seizures, while its blockage of low-voltage gated T-type calcium channels is thought to account for its effect in childhood absence seizures.7
Zonisamide increases GABA-mediated inhibition through two distinct molecular mechanisms, and also acts to reduce extracellular glutamate. Zonisamide has further actions of less significance to epilepsy, such as interactions with dopaminergic and serotonergic pathways implicated in its psychotropic effects.19
Zonisamide, brand name Zonegran, is a new generation antiepileptic drug. It was discovered in 1972, subsequently launched in Japan in 1989, the USA in 2000, and within the EU in 2005. Originally launched by Dainippon Sumitomo Pharma, but taken over by Elan, and then by Eisai.16,19-21
Indications
The European Medicines Agency (EMEA) in Europe and the Food and Drug Administration (FDA) in the USA have approved the use of zonisamide as adjunctive therapy in adults with refractory partial-onset seizures with or without secondary generalisation.22
Japan has a much broader licence including most seizure types, use as adjunctive therapy or monotherapy, and use in adults and children.13
UK/USA off-label use in epilepsy is for a similar profile as the Japanese licence.22 Off-label and experimental uses of zonisamide include for Parkinson’s disease, resting tremor, essential tremor, obesity (eating disorders – particularly binge eating), migraine prophylaxis, and psychiatric disorders.22
Mechanism of Action
Zonisamide is a benzisoxazole-derived sulphonamide anticonvulsant structurally distinct from other AEDs and has multiple mechanisms of action contributing to its effectiveness across seizure types.10,21 Blockage of voltage-gated sodium channels is thought to account for its effect against tonic-clonic and partial seizures, while its blockage of low-voltage gated T-type calcium channels is thought to account for its effect in childhood absence seizures.7
Zonisamide increases GABA-mediated inhibition through two distinct molecular mechanisms, and also acts to reduce extracellular glutamate. Zonisamide has further actions of less significance to epilepsy, such as interactions with dopaminergic and serotonergic pathways implicated in its psychotropic effects.19
Clinical Effectiveness
Twenty-two Phase II/III studies of zonisamide have been conducted in Europe and USA. Four of these were randomised double-blind placebo-controlled multi-centre studies.19
Efficacy criteria for all four were measured as median change in seizure frequency; percentage of participants experiencing 50% or greater reduction in seizures (termed ‘responders’); and adverse effects. Two of the studies also looked at dose-response relationships.19
A 2005 Cochrane review summarised the main results of the four studies as follows:
Twenty-two Phase II/III studies of zonisamide have been conducted in Europe and USA. Four of these were randomised double-blind placebo-controlled multi-centre studies.19
Efficacy criteria for all four were measured as median change in seizure frequency; percentage of participants experiencing 50% or greater reduction in seizures (termed ‘responders’); and adverse effects. Two of the studies also looked at dose-response relationships.19
A 2005 Cochrane review summarised the main results of the four studies as follows:
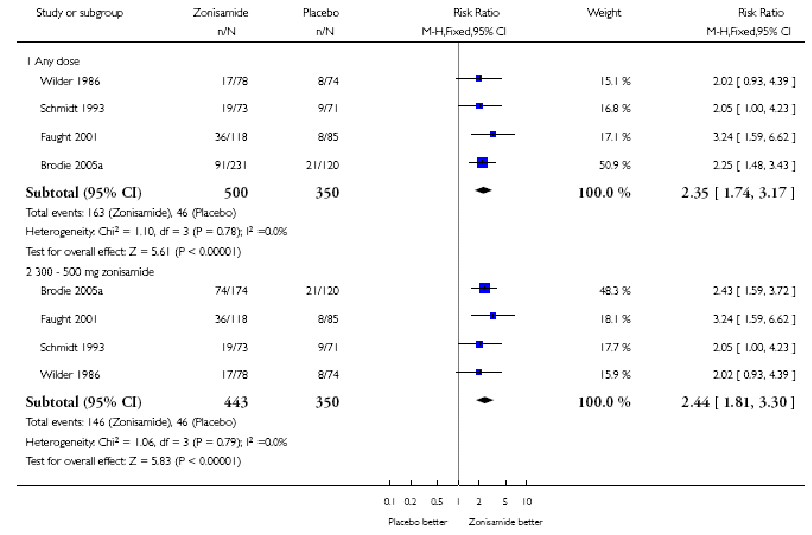
- 850 participants
- 50% reduction in seizure frequency compared to placebo for 300-500mg/day of zonisamide was 2.44 (95% CI 1.81 to 3.30)
- RR for ‘responders’ with any dose zonisamide (100-500mg per day) was 2.35 (1.74 to 3.17)
- RR for treatment withdrawal for 300-500mg/day zonisamide compared to placebo was 1.64 (1.20 to 2.26), and for 100-500mg per day was 1.47 (1.07 to 2.02)5
There are a multitude of Japanese studies on zonisamide, including long-term therapy, use in paediatrics, use as a monotherapy and in generalised epilepsy. Much of the research has been favourable as is reflected in the broad indications approved for zonisamide use in Japan.1
Adverse Events
In line with other AEDs, adverse events occurred more frequently with zonisamide than with placebo. Adverse events experienced tended to be mild to moderate.4 Looking at the four studies together, discontinuation of the medication for any reason was higher with zonisamide than placebo: relative risk of withdrawal 1.47 (95% CI 1.07 to 2.01).5
In general, incidence of severe adverse events was equal in zonisamide and placebo. Renal calculi have been associated in a very small number of cases, likely as a result of the weak carbonic anhydrase inhibition of zonisamide.2,18 Teratogenic potential has been demonstrated in pre-clinical studies and pregnancy is thus a contraindication.13,14,18 Oligohydrosis with secondary hyperthermia in paediatric cases has rarely occurred; this has been reversible with drug discontinuation.2,11,13
It has been demonstrated that tolerance of zonisamide increases along with duration of administration and also with slower initial drug titration. The most commonly experienced adverse events include somnolence, dizziness, agitation, fatigue and ataxia.4 Congruent with other AEDs, CNS-related adverse events are reported early, being most common in the dose-titration phase rather than at steady-state.8,13 Anorexia, with subsequent weight loss, is also associated with zonisamide, and accounts for its use in treating obesity.11
The 2005 Cochrane review of the four studies summarised the relative risk of adverse events as follows:5
Ataxia 4.50 (99% CI 1.05 to 19.22); dizziness 1.77 (99% CI 1.00 to 3.12); somnolence 1.96 (99% CI 1.12 to 3.44); agitation 2.37 (99% CI 1.00 to 5.64); and anorexia 3.00 (99% CI 1.31 to 6.88).5
In February 2009, the US Food and Drug Administration announced that zonisamide has been shown to cause metabolic acidosis and recommended the assessment of bicarbonate levels prior to commencement of therapy and periodically during treatment.17
Pharmokinetics
Zonisamide is rapidly absorbed following oral administration, has a bioavailability of over 95%, and T max (time to maximal concentration) of 2-6 hours (200-400mg). With a stable maintenance dose, a steady state is reached in 14 days.1,19
Zonisamide is hepatically metabolised by way of the cytochrome P450 pathway, specifically CYP3A4. It does not induce its own metabolism and does not induce liver enzymes thus it does not affect the metabolism of concomitant medication, especially as it has a low incidence of protein-binding interactions with other AEDs. Furthermore, zonisamide has no significant effect on the pharmokinetics of the combined oral contraceptive. Zonisamide processing produces no active metabolites.1,10
Plasma half-life is 63 hours, allowing for once daily dosing, although in practice administration is often twice daily when given as an adjunct. The long half-life minimises risk of break-through seizures should a dose be missed.1,22
As a result of CYP3A4 metabolism, co-administration with CYP3A4 inducers or inhibitors may change the pharmokinetic profile of zonisamide. For instance, concomitant administration of phenytoin will reduce the half-life of zonisamide to 27 hours. Note that this will not affect dosing intervals as the half-life is not reduced to below 24 hours.10,22
Adverse Events
In line with other AEDs, adverse events occurred more frequently with zonisamide than with placebo. Adverse events experienced tended to be mild to moderate.4 Looking at the four studies together, discontinuation of the medication for any reason was higher with zonisamide than placebo: relative risk of withdrawal 1.47 (95% CI 1.07 to 2.01).5
In general, incidence of severe adverse events was equal in zonisamide and placebo. Renal calculi have been associated in a very small number of cases, likely as a result of the weak carbonic anhydrase inhibition of zonisamide.2,18 Teratogenic potential has been demonstrated in pre-clinical studies and pregnancy is thus a contraindication.13,14,18 Oligohydrosis with secondary hyperthermia in paediatric cases has rarely occurred; this has been reversible with drug discontinuation.2,11,13
It has been demonstrated that tolerance of zonisamide increases along with duration of administration and also with slower initial drug titration. The most commonly experienced adverse events include somnolence, dizziness, agitation, fatigue and ataxia.4 Congruent with other AEDs, CNS-related adverse events are reported early, being most common in the dose-titration phase rather than at steady-state.8,13 Anorexia, with subsequent weight loss, is also associated with zonisamide, and accounts for its use in treating obesity.11
The 2005 Cochrane review of the four studies summarised the relative risk of adverse events as follows:5
Ataxia 4.50 (99% CI 1.05 to 19.22); dizziness 1.77 (99% CI 1.00 to 3.12); somnolence 1.96 (99% CI 1.12 to 3.44); agitation 2.37 (99% CI 1.00 to 5.64); and anorexia 3.00 (99% CI 1.31 to 6.88).5
In February 2009, the US Food and Drug Administration announced that zonisamide has been shown to cause metabolic acidosis and recommended the assessment of bicarbonate levels prior to commencement of therapy and periodically during treatment.17
Pharmokinetics
Zonisamide is rapidly absorbed following oral administration, has a bioavailability of over 95%, and T max (time to maximal concentration) of 2-6 hours (200-400mg). With a stable maintenance dose, a steady state is reached in 14 days.1,19
Zonisamide is hepatically metabolised by way of the cytochrome P450 pathway, specifically CYP3A4. It does not induce its own metabolism and does not induce liver enzymes thus it does not affect the metabolism of concomitant medication, especially as it has a low incidence of protein-binding interactions with other AEDs. Furthermore, zonisamide has no significant effect on the pharmokinetics of the combined oral contraceptive. Zonisamide processing produces no active metabolites.1,10
Plasma half-life is 63 hours, allowing for once daily dosing, although in practice administration is often twice daily when given as an adjunct. The long half-life minimises risk of break-through seizures should a dose be missed.1,22
As a result of CYP3A4 metabolism, co-administration with CYP3A4 inducers or inhibitors may change the pharmokinetic profile of zonisamide. For instance, concomitant administration of phenytoin will reduce the half-life of zonisamide to 27 hours. Note that this will not affect dosing intervals as the half-life is not reduced to below 24 hours.10,22
Aims
This audit was conducted to evaluate whether, and to what degree, adjunctive zonisamide therapy is efficacious in our patients with refractory partial epilepsy, and how well tolerated it is.
Objectives
Data is to be collected on the following:
Standards
Recommendations for the management of epilepsy advocate monotherapy. If this does not provide satisfactory control of seizures, and alternative monotherapies have been tried, then adjunctive therapy is advised. NICE guidelines do not include zonisamide, however, they do recommend using the new generation AEDs if older ones have failed or if the older ones are unsuitable because:
Nb. The BNF recommendations are the same as those from the EMEA.3,6
Methodology
Retrospective record review of patients on zonisamide.
Identify selected patients, request patient files from patient registry office, collect demographic data, extract relevant information (as stated in objectives), and proceed to collate and interpret data and make appropriate conclusions/recommendations.
This audit was conducted to evaluate whether, and to what degree, adjunctive zonisamide therapy is efficacious in our patients with refractory partial epilepsy, and how well tolerated it is.
Objectives
Data is to be collected on the following:
- Demographics
- Median change in seizure frequency
- Proportion of patients with a 50% or greater reduction in seizure frequency (‘responders’)
- Adverse events: frequency, nature, severity, impact
- Withdrawal: treatment-related or other
- Dose-related response
Standards
Recommendations for the management of epilepsy advocate monotherapy. If this does not provide satisfactory control of seizures, and alternative monotherapies have been tried, then adjunctive therapy is advised. NICE guidelines do not include zonisamide, however, they do recommend using the new generation AEDs if older ones have failed or if the older ones are unsuitable because:
- there are contraindications to the drugs
- they could interact with other drugs the person is taking (notably oral contraceptives)
- they are already known to be poorly tolerated by the individual
- the person is a woman of childbearing potential.12
Nb. The BNF recommendations are the same as those from the EMEA.3,6
Methodology
Retrospective record review of patients on zonisamide.
Identify selected patients, request patient files from patient registry office, collect demographic data, extract relevant information (as stated in objectives), and proceed to collate and interpret data and make appropriate conclusions/recommendations.
Results
This audit included nine patients aged between 23 and 49 (mean age: 37). There were six males and three females. The patients were of various backgrounds and ethnicities. All had been treated with zonisamide for documented indications congruent with guidelines. Dosing titrations were also consistent with guidance.
Zonisamide (ZNS) was withdrawn in 7 cases as it was ineffective. At the time of the audit one of the patients who remained on ZNS had seen no effect with 4 months of use, while the other patient still on ZNS has seen a reduction in seizure frequency from 3 per month to 1 per month over 5 months duration of treatment.
Of the 7 patients who were treated with ZNS and subsequently withdrawn, three received no benefit at all while four experienced an initial seizure reduction which was subsequently lost. It is possible the two patients still on ZNS will follow similar courses.
With only one patient experiencing a 50% or greater reduction in seizure frequency, this audit demonstrates a ‘responder’ rate of 11%. However, this may fall to 0% if the single ‘responder’ (still on ZNS) follows the same course as the others who experienced an initial seizure reduction only to have a subsequent loss of effect.
No median change in seizure frequency has been seen.
Six patients experienced side effects while on ZNS, though causality cannot be established in this instance. The following side effects occurred (with number of patients affected stated in brackets): mild abdominal pain (1), globus pharyngeus (1), terminal insomnia (1), headache (1), light-headedness (1), ataxia (1), and parageusia (1).
This audit included nine patients aged between 23 and 49 (mean age: 37). There were six males and three females. The patients were of various backgrounds and ethnicities. All had been treated with zonisamide for documented indications congruent with guidelines. Dosing titrations were also consistent with guidance.
Zonisamide (ZNS) was withdrawn in 7 cases as it was ineffective. At the time of the audit one of the patients who remained on ZNS had seen no effect with 4 months of use, while the other patient still on ZNS has seen a reduction in seizure frequency from 3 per month to 1 per month over 5 months duration of treatment.
Of the 7 patients who were treated with ZNS and subsequently withdrawn, three received no benefit at all while four experienced an initial seizure reduction which was subsequently lost. It is possible the two patients still on ZNS will follow similar courses.
With only one patient experiencing a 50% or greater reduction in seizure frequency, this audit demonstrates a ‘responder’ rate of 11%. However, this may fall to 0% if the single ‘responder’ (still on ZNS) follows the same course as the others who experienced an initial seizure reduction only to have a subsequent loss of effect.
No median change in seizure frequency has been seen.
Six patients experienced side effects while on ZNS, though causality cannot be established in this instance. The following side effects occurred (with number of patients affected stated in brackets): mild abdominal pain (1), globus pharyngeus (1), terminal insomnia (1), headache (1), light-headedness (1), ataxia (1), and parageusia (1).
Summary of data collected
Outcome or subgroup title (No of patients)
1. 50% reduction in seizure frequency experienced at any time (5)
2. 50% reduction in seizure frequency maintained (1)
3. 50% reduction in seizure frequency not achieved at any time (3)
4. Withdrawal of therapy ( 7)
5. Adverse effects
5.1 mild abdominal pain (1)
5.2 globus pharyngeus (1)
5.3 terminal insomnia (1)
5.4 headache (1)
5.5 light-headedness (1)
5.6 ataxia (1)
5.7 parageusia (1)
Outcome or subgroup title (No of patients)
1. 50% reduction in seizure frequency experienced at any time (5)
2. 50% reduction in seizure frequency maintained (1)
3. 50% reduction in seizure frequency not achieved at any time (3)
4. Withdrawal of therapy ( 7)
5. Adverse effects
5.1 mild abdominal pain (1)
5.2 globus pharyngeus (1)
5.3 terminal insomnia (1)
5.4 headache (1)
5.5 light-headedness (1)
5.6 ataxia (1)
5.7 parageusia (1)
There was no dose-response correlation seen. Two patients experienced drowsiness on increases in doses. These dose-titration phase CNS-related adverse events are described in the literature.
Though seizure frequency reductions were generally not maintained, two patients experienced increased awareness during seizures, and one patient had a reduced duration of seizures. These benefits remained after seizure frequency had returned to pre-ZNS levels.
Conclusions
With nine patients, this audit was relatively small, and so conclusions drawn should be viewed with this in mind. Essentially, zonisamide conferred no effect to half the patients while the other half received an initial benefit in the of form significant seizure frequency reduction followed by a return to pre-ZNS levels and subsequent withdrawal of ZNS therapy.
It is important to note that these are refractory patients – long-term suffers who have trialled most, and in some cases all, of the antiepileptic medications. So perhaps it is unsurprising that another AED is ineffective. Although the Cochrane review demonstrated a favourable ‘responder’ rate this was not reflected by this comparatively small audit. Interestingly, some patients experienced sustaining benefits other than reduced seizure frequency, namely heightened awareness during seizures and reduced duration of seizures. Many of the adverse effects, both dose-related and steady-state, described in the Cochrane review were apparent in our patients.
It would not be prudent to make large conclusions from this audit, though the comprehensive lack of effectiveness of ZNS in this group should be taken into account when considering AED choices for refractory patients in the future. Given the favourable results of the Cochrane review consideration should be given to collaborating with other epilepsy services to establish a larger patient group for further auditing.
On reviewing the patient notes there was no documentation to show monitoring of bicarbonate levels as advised by the FDA in February 2009. This should be considered for future practice.
Though seizure frequency reductions were generally not maintained, two patients experienced increased awareness during seizures, and one patient had a reduced duration of seizures. These benefits remained after seizure frequency had returned to pre-ZNS levels.
Conclusions
With nine patients, this audit was relatively small, and so conclusions drawn should be viewed with this in mind. Essentially, zonisamide conferred no effect to half the patients while the other half received an initial benefit in the of form significant seizure frequency reduction followed by a return to pre-ZNS levels and subsequent withdrawal of ZNS therapy.
It is important to note that these are refractory patients – long-term suffers who have trialled most, and in some cases all, of the antiepileptic medications. So perhaps it is unsurprising that another AED is ineffective. Although the Cochrane review demonstrated a favourable ‘responder’ rate this was not reflected by this comparatively small audit. Interestingly, some patients experienced sustaining benefits other than reduced seizure frequency, namely heightened awareness during seizures and reduced duration of seizures. Many of the adverse effects, both dose-related and steady-state, described in the Cochrane review were apparent in our patients.
It would not be prudent to make large conclusions from this audit, though the comprehensive lack of effectiveness of ZNS in this group should be taken into account when considering AED choices for refractory patients in the future. Given the favourable results of the Cochrane review consideration should be given to collaborating with other epilepsy services to establish a larger patient group for further auditing.
On reviewing the patient notes there was no documentation to show monitoring of bicarbonate levels as advised by the FDA in February 2009. This should be considered for future practice.
References
1. Baulac, M., 2006. Introduction to zonisamide. Epilepsy Research. 68S: S3-S9.
2. Baulac, M., and Leppik, I.E., 2007. Efficacy and safety of adjunctive zonisamide therapy for refractory partial seizures. Epilepsy Research. 75: 75-83.
3. Joint Formulary Committee, 2009. British National Formulary. 57th edition. London: British Medical Association and Royal Pharmaceutical Society of Great Britain.
4. Brodie, M.J., 2004. Zonisamide clinical trials: European experience. Seizure. 13S: S66-S70.
5. Chadwick D.W., and Marson, A.G., 2005. Zonisamide add-on for drug-resistant partial epilepsy. Cochrane Database of Systematic Reviews. Issue 4. Art. No.: CD001416. DOI: 10.1002/14651858.CD001416.pub2.
6. European Medicines Agency, 2008. Annex I – Summary of Product Characteristics [online]. EU, EMEA. Available from:http://www.ema.europa.eu/humandocs/PDFs/EPAR/
zonegran/emea-combined-h577en.pdf [Accessed 12 December 2009].
7. Faught, E., 2004. Review of United States and European clinical trials of zonisamide in the treatment of refractory partial-onset seizures. Seizure. 13S: S59-S65.
8. French, J.A., 2004. Commentary on Faught and Brodie. Seizure. 13S: S71-S72.
9. Fukushima, K., and Seino, M., 2006. A long-term follow-up of zonisamide monotherapy. Epilepsia. 47:1860–1864.
10. Leppik, I.E., 2004. Zonisamide: chemistry, mechanism of action, and pharmacokinetics. Seizure. 13S: S5-S9.
11. Leppik, I.E., 2006. Practical prescribing and long-term efficacy and safety of zonisamide. Epilepsy Research. 68S: S17–S24.
12. National Institute for Health and Clinical Effectiveness, 2004. The diagnosis and management of the epilepsies in adults and children in primary and secondary care [online]. UK, NICE. Available from: http://www.nice.org.uk/nicemedia/live/
10954/29533/29533.pdf [Accessed 12 December 2009].
13. Ohtahara, S., 2006. Zonisamide in the management of epilepsy - Japanese experience. Epilepsy Research. 68S: S25–S33.
14. Ohtahara, S., and Yamatogi, Y., 2004. Safety of zonisamide therapy: prospective follow-up survey. Seizure. 13S: S50-S55.
15. Perucca, E., French, J., and Bialer, M., 2007. Development of new antiepileptic drugs: challenges, incentives, and recent advances. Lancet Neurology. 6: 793–804.
16. Seino, M., 2004. Review of zonisamide development in Japan. Seizure. 13S: S2-S4.
17. US Department of Health and Human Services, 2009. Information for healthcare professionals: zonisamide (marketed as Zonegran, and generics) [online]. USA, Food and Drug Administration. Available from:http://www.fda.gov/Drugs/DrugSafety/
PostmarketDrugSafetyInformationforPatientsandProviders/
DrugSafetyInformationforHeathcareProfessionals/
ucm095251.htm [Accessed 12 December 2009].
18. Vajda, F.J.E., 2007. Pharmacotherapy of epilepsy: New armamentarium, new issues. Journal of Clinical Neuroscience. 14: 813–823.
19. Wilfong, A.A., and Willmore, L.J., 2006. Zonisamide – a review of experience and use in partial seizures. Neuropsychiatric Disease and Treatment. 2: 269–280.
20. Willmore, L.J., 2004. Zonisamide overview of the United States Experience. Seizure. 13S: S57-S58.
21. Yagi, K. 2004. Overview of Japanese experience - controlled and uncontrolled trials. Seizure. 13S: S11-S15.
22. Zaccara, G., and Specchio, L.M., 2009. Long-term safety and effectiveness of zonisamide in the treatment of epilepsy: a review of the literature. Neuropsychiatric Disease and Treatment. 5: 249–259.
Edited for clarity, February 2016.
1. Baulac, M., 2006. Introduction to zonisamide. Epilepsy Research. 68S: S3-S9.
2. Baulac, M., and Leppik, I.E., 2007. Efficacy and safety of adjunctive zonisamide therapy for refractory partial seizures. Epilepsy Research. 75: 75-83.
3. Joint Formulary Committee, 2009. British National Formulary. 57th edition. London: British Medical Association and Royal Pharmaceutical Society of Great Britain.
4. Brodie, M.J., 2004. Zonisamide clinical trials: European experience. Seizure. 13S: S66-S70.
5. Chadwick D.W., and Marson, A.G., 2005. Zonisamide add-on for drug-resistant partial epilepsy. Cochrane Database of Systematic Reviews. Issue 4. Art. No.: CD001416. DOI: 10.1002/14651858.CD001416.pub2.
6. European Medicines Agency, 2008. Annex I – Summary of Product Characteristics [online]. EU, EMEA. Available from:http://www.ema.europa.eu/humandocs/PDFs/EPAR/
zonegran/emea-combined-h577en.pdf [Accessed 12 December 2009].
7. Faught, E., 2004. Review of United States and European clinical trials of zonisamide in the treatment of refractory partial-onset seizures. Seizure. 13S: S59-S65.
8. French, J.A., 2004. Commentary on Faught and Brodie. Seizure. 13S: S71-S72.
9. Fukushima, K., and Seino, M., 2006. A long-term follow-up of zonisamide monotherapy. Epilepsia. 47:1860–1864.
10. Leppik, I.E., 2004. Zonisamide: chemistry, mechanism of action, and pharmacokinetics. Seizure. 13S: S5-S9.
11. Leppik, I.E., 2006. Practical prescribing and long-term efficacy and safety of zonisamide. Epilepsy Research. 68S: S17–S24.
12. National Institute for Health and Clinical Effectiveness, 2004. The diagnosis and management of the epilepsies in adults and children in primary and secondary care [online]. UK, NICE. Available from: http://www.nice.org.uk/nicemedia/live/
10954/29533/29533.pdf [Accessed 12 December 2009].
13. Ohtahara, S., 2006. Zonisamide in the management of epilepsy - Japanese experience. Epilepsy Research. 68S: S25–S33.
14. Ohtahara, S., and Yamatogi, Y., 2004. Safety of zonisamide therapy: prospective follow-up survey. Seizure. 13S: S50-S55.
15. Perucca, E., French, J., and Bialer, M., 2007. Development of new antiepileptic drugs: challenges, incentives, and recent advances. Lancet Neurology. 6: 793–804.
16. Seino, M., 2004. Review of zonisamide development in Japan. Seizure. 13S: S2-S4.
17. US Department of Health and Human Services, 2009. Information for healthcare professionals: zonisamide (marketed as Zonegran, and generics) [online]. USA, Food and Drug Administration. Available from:http://www.fda.gov/Drugs/DrugSafety/
PostmarketDrugSafetyInformationforPatientsandProviders/
DrugSafetyInformationforHeathcareProfessionals/
ucm095251.htm [Accessed 12 December 2009].
18. Vajda, F.J.E., 2007. Pharmacotherapy of epilepsy: New armamentarium, new issues. Journal of Clinical Neuroscience. 14: 813–823.
19. Wilfong, A.A., and Willmore, L.J., 2006. Zonisamide – a review of experience and use in partial seizures. Neuropsychiatric Disease and Treatment. 2: 269–280.
20. Willmore, L.J., 2004. Zonisamide overview of the United States Experience. Seizure. 13S: S57-S58.
21. Yagi, K. 2004. Overview of Japanese experience - controlled and uncontrolled trials. Seizure. 13S: S11-S15.
22. Zaccara, G., and Specchio, L.M., 2009. Long-term safety and effectiveness of zonisamide in the treatment of epilepsy: a review of the literature. Neuropsychiatric Disease and Treatment. 5: 249–259.
Edited for clarity, February 2016.